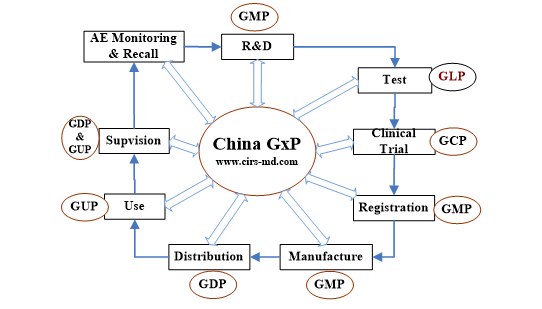
GxP systems are quality management system (QMS) to ensure the safety and effectiveness during the lifecycle of medical product. China has adopted the medical device GxP quality systems these years, it is important to integrate regulatory, market, customer requirements in complying with GxP quality systems in China.
GxP Quality Systems
GxP | |
GMP | The Quality Management Practices for Medical Device Manufacturing 醫(yī)療器械生產(chǎn)質(zhì)量管理規(guī)范 |
GCP | The Quality Management Practices for Medical Device Clinical Trial 醫(yī)療器械臨床試驗(yàn)質(zhì)量管理規(guī)范 |
GDP or GSP | The Quality Management Practices for Medical Device Distributing 醫(yī)療器械經(jīng)營(yíng)質(zhì)量管理規(guī)范 |
GUP | The Quality Management Practices for Medical Device Use 醫(yī)療器械使用質(zhì)量管理規(guī)范 |
GLP | Unimplemented |
How GxP Quality System is Works
How to Comply with Medical Device GxP Quality Systems
GxP Quality systems shall involve in the full product lifecycle. Companies need to know what obligations or regulations to fulfill firstly, then to establish your own appropriate quality management system and follow it.

